Additional Scope of Accreditation for PT Biometrik Riset Indonesia
Writer by Intan Kumala Santriani; Editor by Oktaviani Utami Dewi
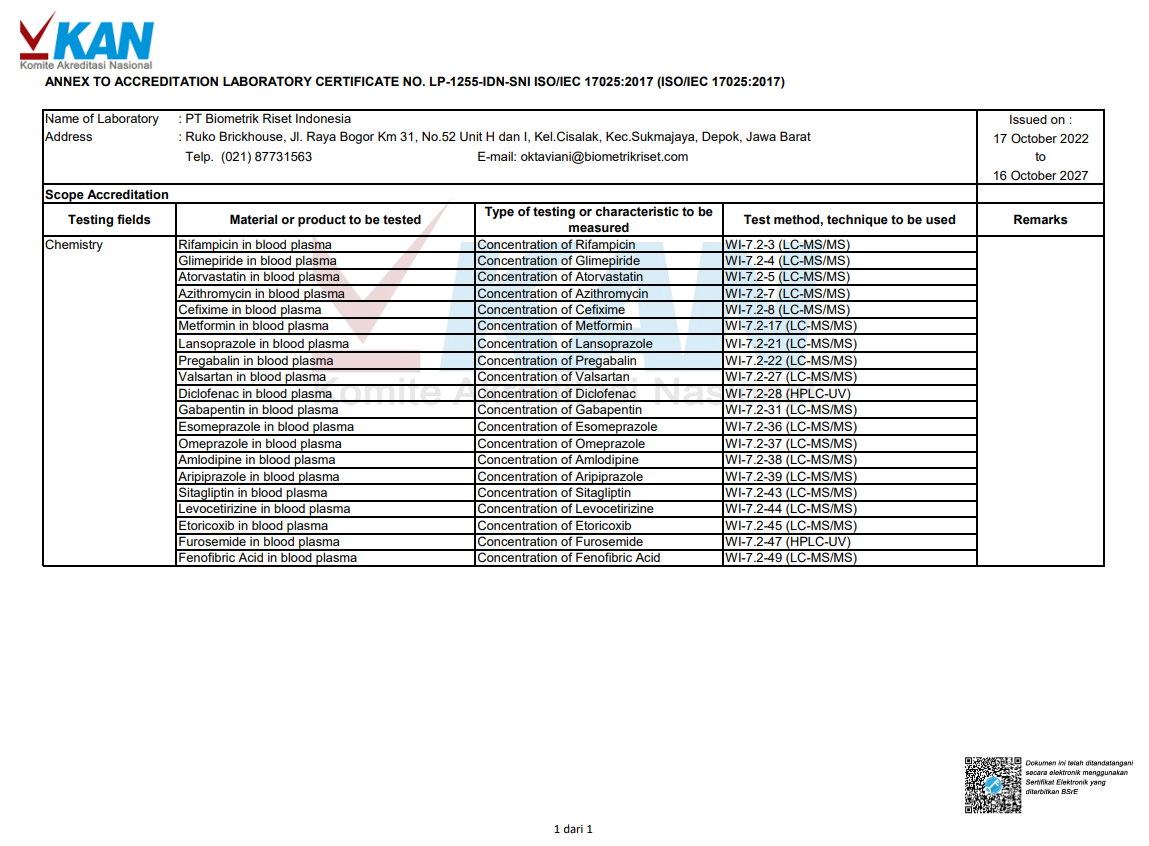
Depok, July 22nd 2022, biometrikriset.com – In line with the issuance of the latest accreditation certificate by the National Accreditation Committee (KAN), the scope accreditation of PT Biometrik Riset Indonesia has also been added. The scope is part of the Testing Laboratory accreditation certificate (LP-1255-IDN-SNI ISO/IEC 17025:2017).

The scope of accreditation has increased by fifteen molecules, from five to twenty testing molecules. The fifth previous molecules is Valsartan, Pregabalin, Lansoprazole, Diclofenac, and Fenofibric Acid. Meanwhile, the fifteen new molecules added to the scope is Rifampicin, Glimepiride, Atorvastatin, Azithromycin, Cefixime, Metformin, Gabapentin, Esomeprazole, Omeprazole, Amlodipine, Aripiprazole, Sitagliptin, Levocetirizine, Etoricoxib, and Furosemide.
“By increasing the scope of accreditation, it will also increase the access and trust of sponsors to conduct bioequivalence (BE) studies in Biometrik. However, BE studies can be carried out in Biometrik outside of the molecule that is within the scope of accreditation as long as there is method validation for the molecule to be tested for BE study. Biometrik has more than 60 validation methods that have been validated with valid results and meet the parameters required in the European Medicines Agency (EMA) guidelines” said Mrs. Effi as Director of PT Biometrik Riset Indonesia.
Leave a comment